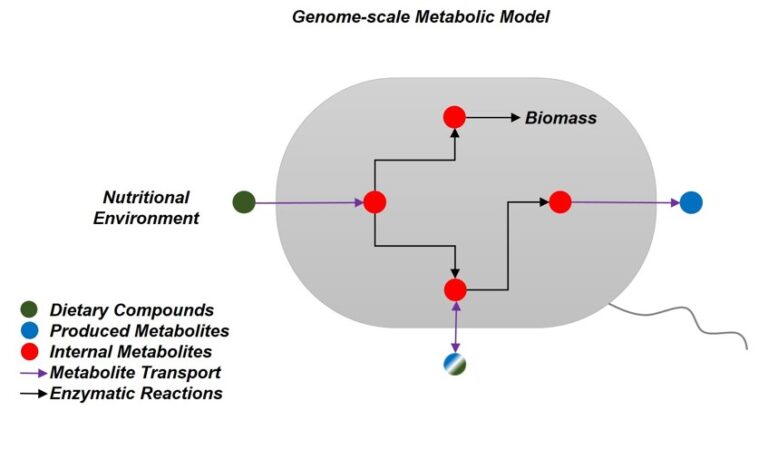
Core project INF (PIs Franke, Höppner) aims to provide support in data management and analysis. The project will offer the necessary resources and expertise to ensure good scientific practice in data handling and processing. It will adapt the existing infrastructure to technology platforms newly added to the CRC, including microscopy (Z2) and metabolomics (Z3). The project serves as the main contact point for training in data analysis, CRC-overarching analysis and data integration. Additionally, it will further expand the recently developed public CRC data platform and engage in planning the CRC data legacy.
INF
Science Support
Data management and bioinformatics support
INF
Researchers
Researchers
INF
INF
Associated Postdoctoral Researcher