Smelly sludge: A happy place for worms and microbes
Kiel research team uses near-natural compost mesocosm experiment to demonstrate that worm hosts and the associated microorganisms jointly mediate adaptation to a novel environment
All multicellular organisms – from the simplest animal and plant organisms to humans – live in close association with a variety of microorganisms, known as the microbiome, which colonise on and in their tissues and form symbiotic relationships with the host. Many vital functions such as nutrient uptake, regulation of the immune system or even neurological processes result from the interactions between host organism and microbial symbionts. The functional cooperation between host and microorganisms, which scientists refer to as a metaorganism, is being studied in detail at Kiel University in the Collaborative Research Center (CRC) 1182 “Origin and Function of Metaorganisms”.
Scientists assume that the microbiome can contribute significantly to the environmental adaptation of an entire organism. This may be due to the rapid adaptability of microorganisms, which can react many times faster to changing environmental conditions than their host organism. Numerous studies have already shown that the microbiome responds to changing environmental factors such as higher temperatures, for example, by changing its species composition and thus contributing to the environmental adaptation of the host organism. However, many of these studies have so far focused only on the microbiome, but not the host.
Researchers from the Evolutionary Ecology and Genetics group led by Professor Hinrich Schulenburg at the Institute of Zoology at Kiel University have now focused on the joint influences of host organisms and the microbiome in a new study. To this end, they developed an innovative cultivation method to investigate the adaptations of a metaorganism to novel environmental conditions: The nematode Caenorhabditis elegans evolved in a long-term experiment over 100 days together with an originally specified selection of microorganisms under complex and near-natural environmental conditions in a compost habitat. The researchers then analysed changes both on the part of the hosts and in the microbiome and found that under certain conditions both contribute jointly to the optimal adaptation of the whole organism to a new environment. The Kiel researchers, who cooperated in this study with Professor Brendan Bohannan and his team from the University of Oregon in the USA, recently published their results in The ISME Journal.
Smelly compost allows insights into the course of environmental adaptation
Thanks to its very short generation time of only about three and a half days, C. elegans is particularly suitable for studying mechanisms of evolution. Earlier research, to which Schulenburg’s research group contributed, identified the worm’s natural microbiome or characterized certain microbiome functions such as pathogen defence. However, it is generally unknown how host and microbiome evolve together over longer time spans and thereby respond to variable factors in their natural environment. To address this aspect, the Kiel research team investigated C. elegans and its microbiome over a long period of time under near-natural conditions.
“In nature, the worm lives on decaying organic material such as fruit and vegetable matter. We tried to recreate such a compost environment under controlled laboratory conditions and developed so-called mesocosms based on plant material. With the help of these artificial compost habitats, we succeeded in creating a sustainable habitat for the worm and its microbiome. In the meantime, the worms have been surviving continuously for almost two years in this environment, which is particularly disgusting to the human eye,” explains Dr Carola Petersen, one of the first authors and a research associate in the Schulenburg group.
Host and microbiome can contribute together to environmental adaptation
From this laboratory compost, the research team isolated C. elegans and samples of its microbiome after 100 days – equivalent to about 30 worm generations They then conducted various experiments in a so-called common garden approach to determine the effects of microbiome composition and host genetics on the adaptation of the metaorganism. “To do this, we brought together original and evolved worms and microbes in all possible combinations and cultivated each in the near-natural environment in new mesocosm compost boxes and also on agar plates. We then used various parameters to determine the fitness of the metaorganism as a whole,” explains CRC 1182 member Petersen.
“Especially striking were the high fitness values of the worms in one particular box, whose population thrived in extraordinary numbers compared to the other experimental replicates. These worms showed a strong deviation in gene expression compared to the original population, even though they had the same original microbiome,” Petersen emphasises. This fitness advantage is therefore genetically controlled and thus an environmental adaptation. The advantage for the organism as a whole appears to be manifested in an improved stress response, which assists the worms to cope with complex environments such as the compost habitat with its numerous structural and physiological challenges.
In addition, the researchers were able to prove that the evolved microbiome also contributed to the worm’s fitness advantages: “The clear effect of increased population growth was only seen in the compost environment in the presence of the adapted microbes, but not on agar plates. So this component also exerts a highly specific influence on the fitness of the worm population,” Petersen continues. Overall, the study thus provides experimental evidence in the form of specific changes in the microbiome composition and also in the gene expression of the nematodes, showing that adaptation to a new environment can be jointly influenced by host and microbiome.
Novel model system allows quantification of the influences of host genetics and microbiome
“These environmental adaptations are often based on quantitative genetic changes. How exactly the microbiome interacts with the host’s genetics to improve joint fitness has not yet been studied in detail,” says Schulenburg, vice spokesperson of the CRC 1182. “With our novel mesocosm system, we have for the first time created reproducible conditions that allow us not only to confirm the contributions of host and microbiome to this adaptive process, but also to quantify the involved mechanisms and processes in the future,” Schulenburg adds. To this end, the CRC 1182 researchers want to extensively analyse the genetic data of worms and microbes obtained from the long-term experiment in further research to clarify in detail the influences of host and microbial genes on evolutionary adaptation.
Original publication:
Carola Petersen, Inga K. Hamerich, Karen L. Adair, Hanne Griem-Krey, Montserrat Torres Oliva, Marc P. Hoeppner, Brendan J.M. Bohannan, Hinrich Schulenburg (2023): Host and microbiome jointly contribute to environmental adaptation. The ISME Journal
First published: 06. September 2023 https://doi.org/10.1038/s41396-023-01507-9
Images are available for download:
www.uni-kiel.de/de/pressemitteilungen/2023/220-petersen-isme-authors.jpg
Caption: Hanne Griem-Krey, Dr Carola Petersen and Inga Hamerich (from left to right), together with their colleagues demonstrated in the new study that both host and microbiome can contribute to the environmental adaptation of a metaorganism.
© Christian Urban, Kiel University
www.uni-kiel.de/de/pressemitteilungen/2023/220-petersen-isme-mesocosms.jpg
Caption: In the near-natural compost mesocosms (shown here after 0, 100 and 600 days, from top to bottom), the researchers were able to observe the adaptation of host organisms and microorganisms to a complex environment over a long period of time.
© Dr Carola Petersen
www.uni-kiel.de/fileadmin/user_upload/old_news_images/2016/2016-152-2.jpg
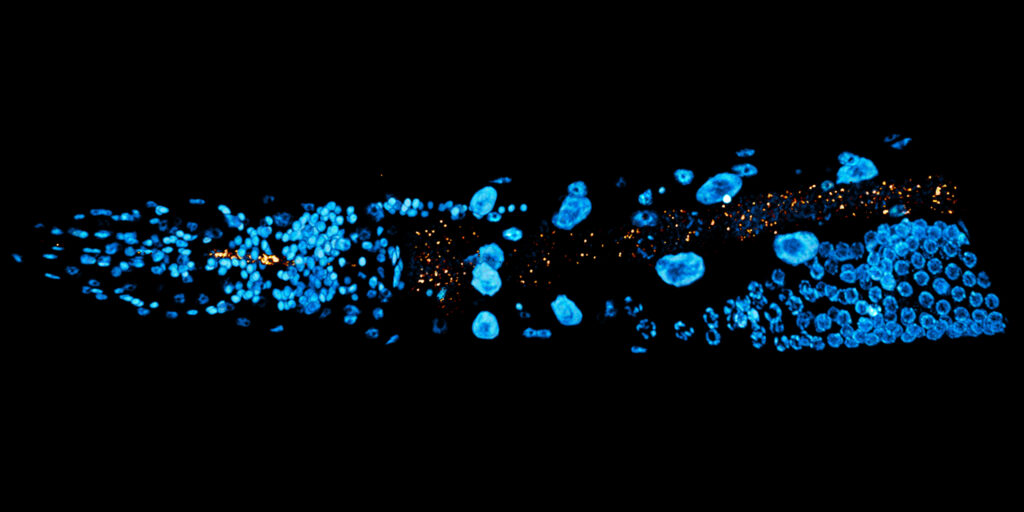
Caption: The nematode C. elegans and its microbiome (shown here in red) form an ideal model system to study the effects of microbiome composition and host genetics on the adaptation of the metaorganism.
© Dr Philip Dirksen
Contact:
Prof. Hinrich Schulenburg
Evolutionary Ecology and Genetics group, Head
Zoological Institute, Kiel University
Phone: 0431-880-4141
Email: hschulenburg@zoologie.uni-kiel.de
More information:
Evolutionary Ecology and Genetics group,
Zoological Institute, Kiel University
www.uni-kiel.de/zoologie/evoecogen